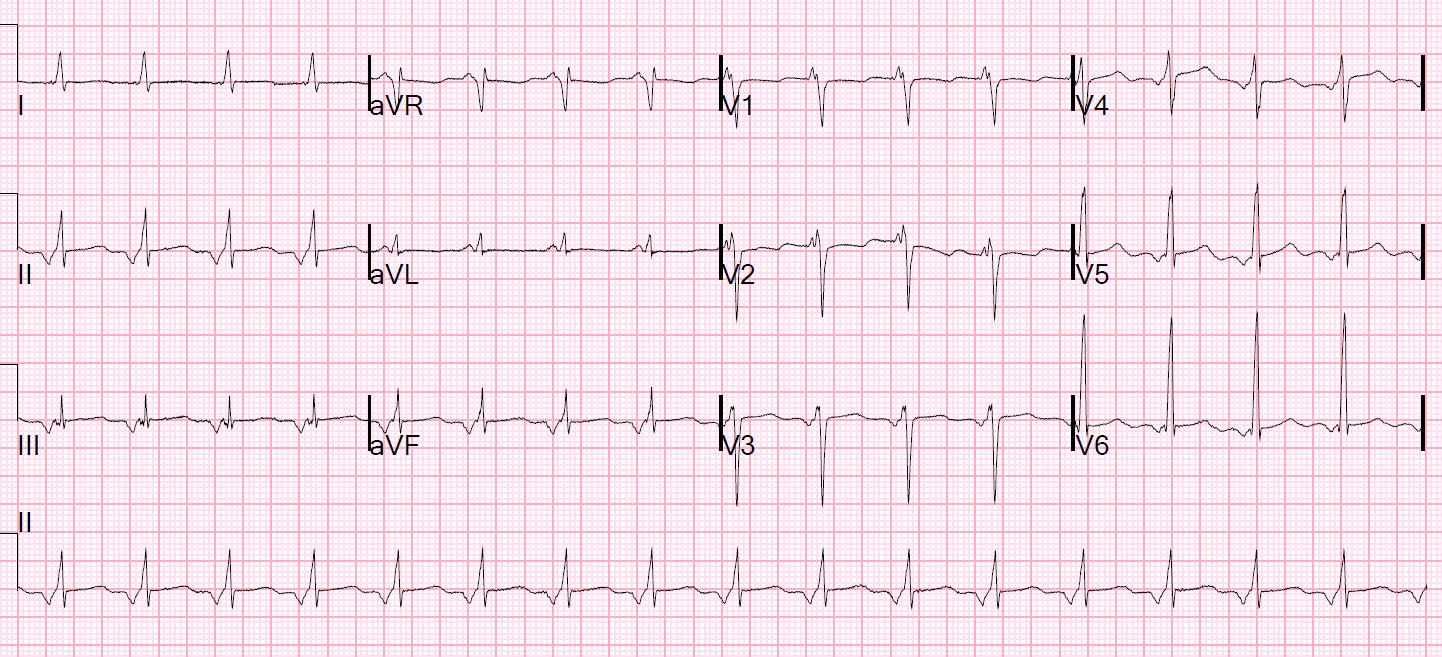

We conducted whole-exome sequencing for 124 patients with polymicrogyria and identified de novo ATP1A3 variants in eight patients. Polymicrogyria is a common malformation of cortical development whose etiology remains elusive. Patients with AHC should be evaluated to identify risk of sudden death. ATP1A3 variants may represent an independent cause of sudden unexplained death. Patients with AHC who carry the ATP1A3‐D801N variant have significantly shorter QTc intervals and an increased likelihood of experiencing bradycardia associated with life‐threatening arrhythmias. Rare variants in ATP1A3 were identified in a large cohort of genotype‐negative patients referred for arrhythmia and sudden unexplained death. Two patients with the ATP1A3‐D801N variant experienced ventricular fibrillation, resulting in death in 1 patient. ATP1A3 genotype status was significantly associated with shortened QTc by multivariant regression analysis. The mean QTc for ATP1A3‐D801N was 344.9 milliseconds, which varied little with age, and remained <370 milliseconds throughout adulthood. QTc measurements were significantly shorter in patients with positive ATP1A3 variant status ( P <0.001) than in patients with genotype‐negative status, and significantly shorter in patients with the ATP1A3‐D801N variant than patients with other variants ( P <0.001).
IVCD INTRAVENTRICULAR CONDUCTION DELAY MANUAL
Manual remeasurements of QT intervals and QTc calculations were performed by 2 pediatric electrophysiologists. Patients were grouped according to AHC presentation (typical versus atypical), ATP1A3 variant status (positive versus negative), and ATP1A3 variant (D801N versus other variants). We analyzed 12‐lead ECG recordings from 62 patients (male subjects=31, female subjects=31) referred for AHC evaluation. The objective was to determine the role of ATP1A3 genetic variants on cardiac outcomes as determined by QT and corrected QT (QTc) measurements. Although these patients experience a high rate of sudden unexpected death in epilepsy, the pathophysiologic basis for this risk remains unknown. Pathogenic variation in the ATP1A3 ‐encoded sodium‐potassium ATPase, ATP1A3, is responsible for alternating hemiplegia of childhood (AHC). 2,31 Studies focusing on the D801N variant show through homology modeling that the D801N variant creates a positive dipole which consequently affects the flow of potassium through the pump via electrostatic repulsion 41 and reduced sodium-potassium ATPase activity and pump current in mammalian cells. 30,31, Although ATP1A3-D801N is associated with a mild-moderate form of AHC from a neurological perspective, 21,40 it has been implicated in more severe cardiac phenotypes. Previous work in principally neuronal models has revealed that differing protein variants in the alpha-3 subunit of the ATP1A3 gene that result in the syndrome of cerebellar ataxia, areflexia, pes cavus, optic atrophy, and sensorineural hearing loss rapid-onset dystonia parkinsonism and AHC cause specific disruptions that cause reduced affinity at the cytoplasmic surface, widely distributed protein dysfunction, or dysfunction at the cytoplasmic surface in the transmembrane domain, respectively.
